health /
What is the molecular geometry of nhcl2
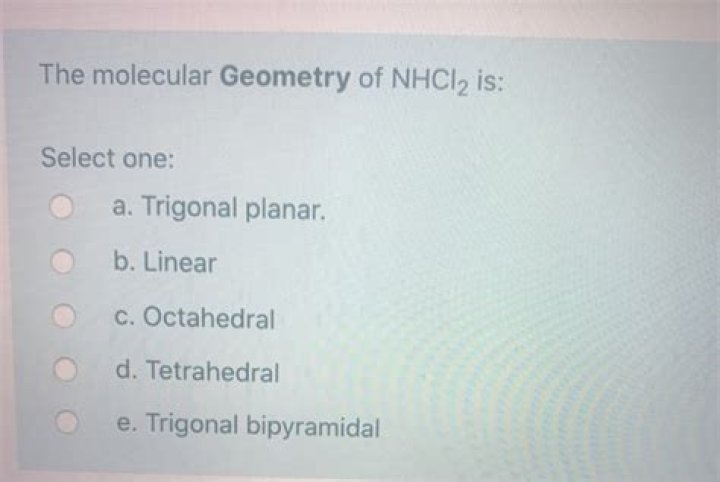
Is NHCl2 a polar molecule?
The “N-Cl” bond also is polar because chlorine is more electronegative than nitrogen. … The trigonal pyramidal geometry means that the vectors will not cancel out, so chloramine is a polar compound.How do you find the molecular geometry?
Steps Used to Find the Shape of the Molecule- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
What is the molecular geometry for bf3?
trigonal planar The geometry of the BF 3 molecule is called trigonal planar (see Figure 5). The fluorine atoms are positioned at the vertices of an equilateral triangle. The F-B-F angle is 120° and all four atoms lie in the same plane.Is NHCl2 a hydrogen bond?
NHCl2 has hydrogen bonding becauseof the presence of N-H bond in addition to London forces. (b) Due to presence of hydrogen bonding which is a strong bond compared to dipole-dipole and London forces, NHCl2 is most likely to be in comdensed phase.What are the types of molecular geometry?
Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the VSEPR theory.What is the molecular geometry of h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.What is the boiling point of nhcl2?
It is a colorless liquid at its melting point of −66 °C (−87 °F), but it is usually handled as a dilute aqueous solution, in which form it is sometimes used as a disinfectant. Chloramine is too unstable to have its boiling point measured.…
Monochloramine.
| Names | |
|---|---|
| show SMILES | |
| Properties | |
| Chemical formula | NH 2Cl |
| Molar mass | 51.476 g mol−1 |
Is Dichloramine unstable?
Dichloramine is a reactive inorganic compound. It has the formula NHCl2. The yellow gas is unstable and reacts with many materials. It is formed by a reaction between ammonia and chlorine or sodium hypochlorite.Does no2 have hydrogen bonding?
In scenario III, diamond only has weak van der Waals interactions whereas nitric oxide can participate in dipole-dipole interactions as well (note that nitric oxide doesn’t have hydrogen atom and, therefore, cannot participate in hydrogen bonds). This means that molecule B has the higher boiling point in scenario III.What is Monochloramine water?
Monochloramine is a disinfectant used to eliminate bacteria and other microbes as a part of drinking water treatment. While chlorine is the most commonly used primary disinfectant, an increasing number of water providers are using monochloramine to help them comply with new regulations.What is the molecular geometry of butane?
Tetrahedral Properties of Butane| Name of Molecule | Butane |
|---|---|
| Molecular Geometry | Tetrahedral |
| Hybridization | sp3 |
| Molecular Formula | C4H10 |
| Molecular Weight | 58.12 g/mol |



